Life Science and Medical
When a sample can "run away" from laser light we internally call it “Biophotonics”.
Fun aside. Laser light has made significant contributions in understanding the Science of Life in the last decades. It also has had important roles in diagnosis, real-time monitoring, guiding therapy, and enabling new therapeutic strategies.
Illuminating the functionality of life with the precision of light
Light has transformed the way we see life itself. In modern biomedical research and clinical diagnostics, lasers are at the heart of innovation — driving breakthroughs from fundamental cell imaging to functional understanding. At TOPTICA, we provide laser technology that empowers scientists, engineers, and clinicians to explore biological systems with unmatched imaging quality, chemical sensitivity, 3D resolution and multi-modal flexibility.
The power of lasers in life sciences
Lasers are the cornerstone of optical imaging and analysis in life sciences. Their unique combination of wavelength, brightness, tunability, and temporal control enables researchers to visualize structures and processes at the sub-cellular and even molecular level. Whether it’s revealing intricate neural networks or tracking single molecules, photonic solutions deliver the illumination that makes discovery possible.
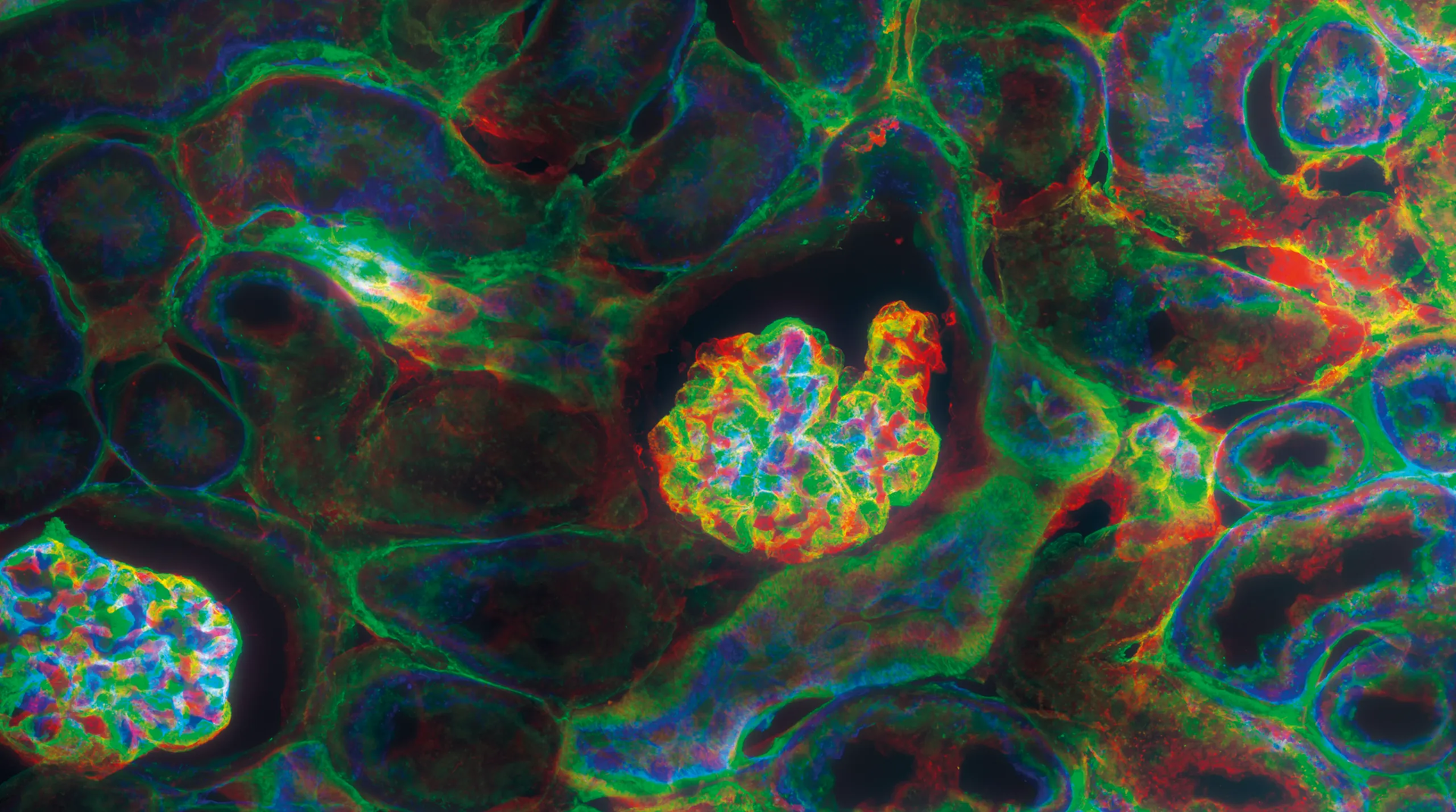
In confocal microscopy (LSCM), tightly focused laser beams scan biological specimens point by point, generating high-contrast optical sections with exceptional spatial resolution. Our stable, low-noise laser sources with direct modulation of all wavelengths ensure optimal excitation efficiency, no optical cross-talking and minimal photobleaching — key for long-term imaging in live-cell studies.
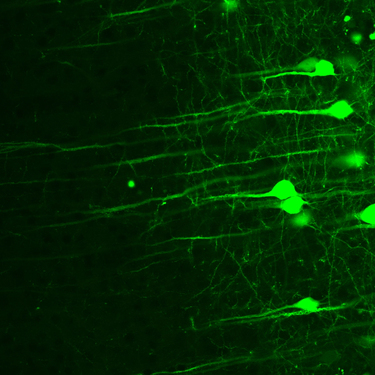
High peak power and a clean pulse shape are necessary for directly visualizing neurons and neuronal activity in vivo. Femtosecond lasers enable precise optical stimulation and recording, revealing the secrets of brain function and connectivity.
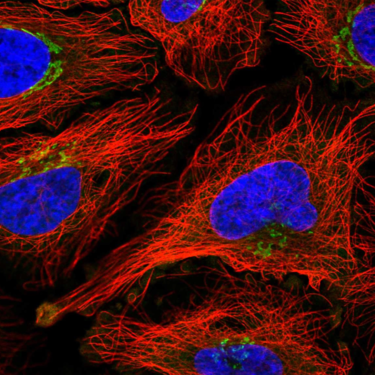
Two-photon microscopy combines the precision of laser scanning with the power of nonlinear optics to visualize subcellular structures in depth, in three dimensions, and in real time. Our femtosecond pulsed lasers deliver the high peak power and wavelength flexibility necessary for this powerful technique.
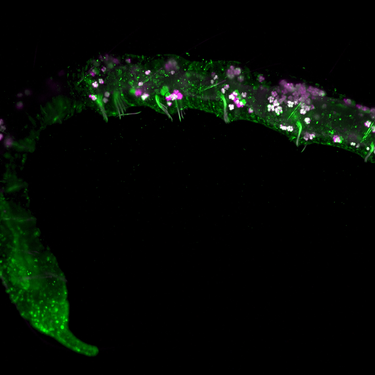
Light sheet microscopy (SPIM) takes optical sectioning to the next level by illuminating samples with a thin sheet of light, reducing phototoxicity while achieving fast 3D imaging. With our multi-line laser platforms, researchers can simultaneously excite multiple fluorophores to capture rich volumetric data of developing embryos, organoids, and living tissues.
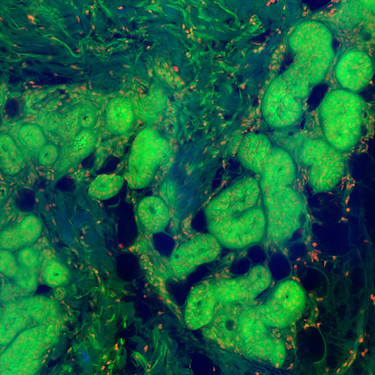
Pushing beyond the diffraction limit, super-resolution techniques such as STED, SIM, PALM, and STORM demand lasers with exceptional stability, pulse control, high power levels and spectral purity. Our systems provide the timing accuracy and power consistency required to achieve nanometer-scale insights into molecular architecture.
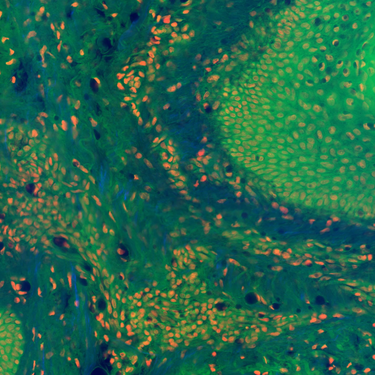
In ophthalmology, our femtosecond lasers support cutting-edge surgical systems — from corneal surgery (LASIK, SMILE) to enhanced cataract surgery, surgery and future potential like Presbyopia and glaucoma innovations — combining precision illumination with patient safety and comfort.

In life sciences and medical research, light is key to understanding the complexity of living systems. Today, advanced laser-based technologies allow scientists and clinicians to visualise molecular and structural information without the use of dyes or contrast agents. This technique reveals the native biochemical composition, metabolism, and morphology of tissues, cells, and biomolecules, providing real-time insights with minimal sample preparation and no phototoxicity.